Bioenergetics is the scientific study concerned with the flow of energy among organisms at cellular level and is based upon the principles of biochemistry and molecular biology.
Energy production in organisms involves certain processes ranging from photosynthesis in plants and that of oxidation metabolism in other organisms like human. Therefore, energy is the essential requirement of all living organisms to carry out their body functions. Cells produce energy and utilize it while maintaining a balance and equilibrium of energy in cell and body of organism.
Type of energy in living body
Two types of energy involved in cellular reactions of living beings:
1. Kinetic energy
Energy that is related to movement and motion is kinetic energy. For instance, light and heat energy.
2. Potential energy
This is stored form of energy such as the one stored in chemical bonds. Moreover, the breaking of these bonds release a substantial amount of energy . Basically, energy is produced at the small molecular level by breakage of phosphate bonds in adenosis triphosphate molecule-. This molecule is a basic reservoir of energy conservation in cells. Some other organisms may produce other energy rich compounds in the cells. The breaking of high energy phosphate bond in ATP molecule yield free energy in cell for various cellular functions.
The flow of energy is often between a system and its surroundings. Three types of system are there regarding energy flow and exchange system.
- Open system—that involves exchange of both energy and matter with the surroundings
- Closed system—that involves only exchange of energy with outside
- Isolated system—that does not exchange matter or energy with its surroundings
So, bioenergetics involve the aspects how cell produces energy and maintain its flow in system.
History of Bioenergetics
Science of bioenergetics started in 17th century by the work of J.Priestly and Antonio Lavoisier.
In 1940’s complete description of glycolysis (redox reaction) was proposed by Embden and Mayerhof mutually. Similar contributions were done by discovery of kreb cycle and ATP production mechanism- chemiosmosis and many other enzymes like NAD- Nicotinamide adenine dinucleotide, cytochrom complexes that are essential part of these energy producing processes.
Similarly, mitochondrial bioenergetics basically gained importance in 20th century by work of Leninger, Racker and others which was based upon the “Chemical coupling hypothesis” of energy flow in cells that described energy conservation mechanism in cells. Bioenergetics study describes the energy pathways at three stages:
- Growth
- Development
- Metabolism
Growth is the reproduction of cells of organisms. Development involves all the energy changing steps and energy flow pathways between body of organisms and the surroundings.
What is metabolism?
The term metabolism implies to collaborative processes of energy production and its usage in other functions of cell. Actually, it is the sum of all processes occurring in cell to maintain equilibrium of energy in it.
Metabolism includes:
- Catabolic reaction
- Anabolic reactions
1. Catabolic reactions
Breakdown of complex e molecule into simpler ones yield free energy in cells and the process is termed as the catabolic reaction. For example, glycolysis which is the breakdown of sugar (energy rich compound) to simpler substances that yields energy for cellular functions. The ATP molecule is the energy reservoir of cell.
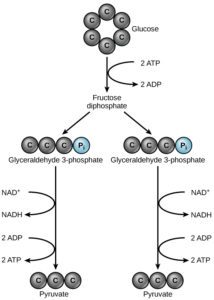
Glycolysis is the step by step and oxidative breakdown of glucose (energy rich compound) to simpler substances like pyruvate that yield energy in the form of ATP & NADH. It is a catabolic and exergonic reaction of living cell system.
2. Anabolic reactions
On the other hand the anabolic reactions are those that involve formation of complex molecules by joining the simpler ones. This process requires energy of carbolic reactions. For example, protein synthesis by bonding of amino acids in chain structure. The protein synthesis by joining of large number of amino acids together is an anabolic reaction. Similarly, the monosaccharide join together by glycosidic linkage to form disaccharides. These reaction require and utilize the energy from catabolic reactions.
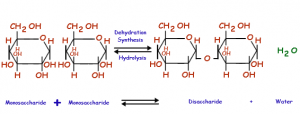
Coupling reactions of catabolism and anabolism
Both anabolic and catabolic pathways are coupled. Metabolism includes both catabolic and anabolic reactions that maintain the energy of system constant.ATP is the principle energy storing compound in cell. The anabolic reaction utilize the energy of catabolic reactions. Thus, both coupling reaction maintain an equilibrium of energy.
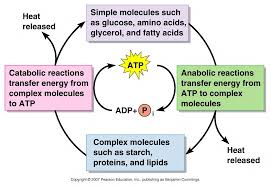
Bioenergetics and Thermodynamics
Bioenergetics implies the principles of biochemistry and physics and also follows the laws of thermodynamics. Thermodynamics defines as:
“The study of energy transformations in cells of organisms and also in physical systems is thermodynamics.”
Laws of thermodynamics
- Firstly, energy of the system can neither be created nor can it be destroyed but can be changed from one form to other. In other words the energy of system remains constant.
- Secondly, the entropy of the system always tends to increase. Entropy is the disorder of randomness of system.
Free energy of a system is the total amount of energy of that system. Therefore, bioenergetics or the energy changes of the system are given as:
![]()
- Where, G is Gibbs unit of free energy
- H is enthalpy of system
- T is absolute temperature
- S is the change in entropy o system
Types of reaction in cellular functions
Two types of reactions are involved in cellular functions:
- Exergonic reactions
- Endergonic reactions
Exergonic reactions
The type of reaction which proceeds with the releases of some free energy is termed as the exergonic reactions. For example, catabolic reactions. In this case, the free energy of system ∆G is negative.
Endergonic reactions
The reaction which proceeds with usage of energy that is absorbed from surroundings is termed as the endergonic reactions i.e anabolic reactions. In this case, ∆G is positive.
The energy used in endergonic reactions comes from the exergonic reactions that release it. This is how, both are coupled reactions in cell system.
Bottom line
The discipline of bioenergetics helps a lot in understanding the energy transformations in living system. Energy releasing reactions and those which are energy requiring have close association and coupling to maintain the energy equilibrium of cell.