Enzyme specificity describes the tendency of an enzyme to catalyze a specify biochemical reaction by binding to specific substrate. Enzymes are specific for the type of reaction that they catalyze.
For example, the digestive enzymes break down the macromolecular food particle into small digestible particles. Trypsin enzyme catalyzes the breakdown of proteins. Amylase enzyme catalyzes carbohydrate digestion.
What are enzymes?
Enzymes are the biological catalysts that speed up the chemical reactions in cells of living organisms. Enzymes are proteins having a 3D structure that needs to be maintained for an enzyme to remain functional. They lower thee activation energy of biochemical reaction in cells and thus speed up the rate of reaction. Activation energy of enzyme is the amount of energy required to initiate a reaction. With the help of enzymes, the substrates are converted into products that are utilized by the cell. For example, the digestive enzyme protease breaks down the large protein molecules into smaller digestible peptides.
Enzymes are proteins and each enzyme has a specific structure consisting of active site and a catalytic site. Active site is the point of attachment of substrate with the enzyme that has a specific amino acid sequence. The catalytic site of enzyme controls its enzymatic activity. In addition to these sites, some enzymes may require a co-factor (inorganic components or some metal ions) or a co-enzyme that are organic part of enzyme
Mechanism of enzyme action
The sub stare attaches itself with the enzyme at its active site by means of hydrophobic/hydrophilic or some other type of intramolecular interactions. The attachment of enzyme with the substrate forms an enzyme –substrate complex that acts as transition state and is unstable. This enzyme –substrate complex breaks into products and the enzyme is again free to catalyze another reaction. The binding of substrate occurs on the basis of its shape, charge and the type of interactions formed between the substrate and enzyme.
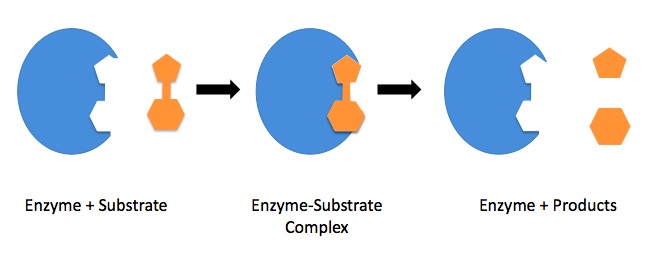
The Lock And Key Model of enzyme suggests that the enzyme has a definite rigid structure that cannot be changed. While the Induced Fit Model of enzymes suggest that the enzyme’s active site is a flexible structure to some extent and may changes its configuration to allow binding of substrate molecules with the enzyme. So the induced fir model is more acceptable to explain mechanism of enzymes action.
Enzyme specificity
This describes the tendency of an enzyme to catalyze a specify biochemical reaction by binding to specific substrate. Enzymes are specific for the type of reaction that they catalyze.
For example, the digestive enzymes break down the macromolecular food particle into small digestible particles. Trypsin enzyme catalyzes the breakdown of proteins. Amylase enzyme catalyzes carbohydrate digestion.
For the proper functioning of enzyme, its specificity is one of the important factors. Some enzymes may catalyze more than one type of reaction their catalysis activity will be affected. So the type of enzyme is specific for specific reaction catalysis.
How enzyme specificity works?
Enzyme specificity is achieved on the basis of its 3D structure, the type of amino acid sequence that makes its active site, type of substrate and most importantly the type of interactions (hydrophobic/hydrophilic etc) that are formed between enzyme’s active site and substrate molecule. The amino acid groups of the active site of enzyme may be of different type so it contributes in determining the specificity of enzyme. For example,
- Some amino acid groups that give conformational changes to enzyme-substrate complex
- Orientation groups that provide specific orientation for substrate attachment
- Groups that provide affinity for developing intramolecular interactions between enzyme and substrate
- Catalytic groups that create enough tension to provide bond breaking strength to enzymes
Types of enzyme specificity
- Substrate/ Absolute specificity
- Bond/ Relative specificity
- Linkage specificity
- Group specificity
- Optical specificity
- Geometrical specificity
- Co-factor specificity
1. Substrate/ absolute specificity
The specificity of enzymes for their substrate is absolute specificity. Enzymes not only show absolute specificity for their substrate but also for the type of reaction they catalyze.
For example, the β-1,4-glycosidic linkage of lactose can only be hydrolyzed by lactase enzyme and this reaction yields glucose and galactose.
The α-1,4-glycosidic bond present between two glucose residues in maltose can only be hydrolyze by maltase enzyme.
Succinate dehydrogenase specifically binds to succinate in Kreb cycle. Glutamate is specific substrate for L-glutamate dehydrogenase.
2. Bond specificity
Also termed as “Relative Specificity” of enzymes. Enzymes are specific for structurally related molecules. These structures share closely related bonding pattern. For example, the peptide bond, glycosidic bond and ester bond etc. Due to similar bonds present in these structures, the enzyme can bind and catalyze structurally related molecules.
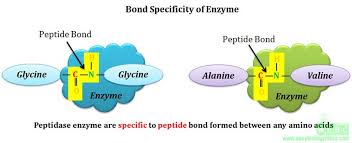
Representation of bond specificity of enzyme that hydrolyze the peptide bond in substrate molecule
For example, the ability of α-amylase enzyme to hydrolyze 1-4-glycosideic linkage in glycogen and starch. In this case, the enzyme only considers the glycosidic bond not the substrate. Same is the case for lipase enzyme that breaks ester bond both in fatty acids and glycerol. The L and D forms of amino acids are structurally related to each other so they also have the ability to act as substrate for same enzyme.
3. Group specificity
This is the moderate type of enzyme specificity as the enzyme considers the bonds present in substrate molecule and the surrounding groups of bond. The class of enzyme showing group specificity is peptidase class that hydrolyzes peptide bond in substrate molecule. Peptidase class includes both the endo and exopeptidases.
Pepsin is one of the digestive enzymes. The chief cell of stomach produce and release it and its function is to hydrolyze the peptide bond. And for this the amino group is an aromatic amino acid such as tryptophan, tyrosin, phenylalanine etc.
One other example is chemotrypsin enzyme that is a constituent of pancreatic juice and it also hydrolyzes the peptide bond in which an aromatic amino acid contributes its carboxylic group as the surrounding group of peptide bond.
Carboxypeptidase and aminopeptidase hydrolyze the peptide bonds of substrate protein at its C-terminal and N-terminal residue.
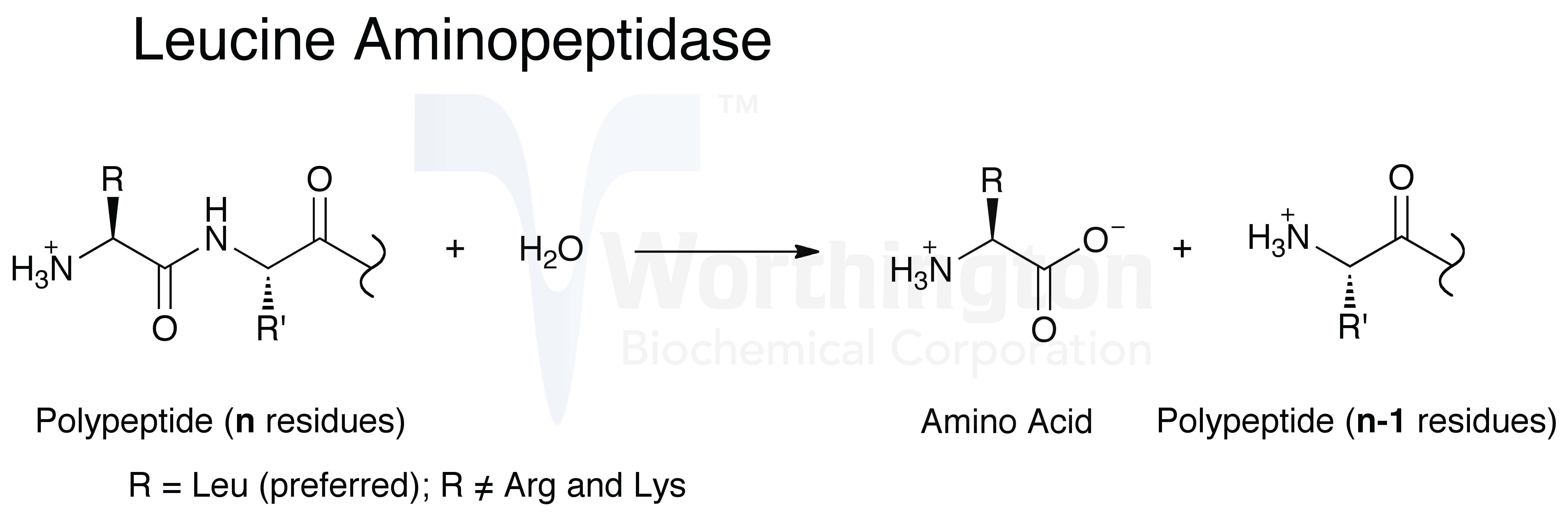
“Aminopeptidase enzyme hydrolyzes peptide bond at N-terminal of protein.”
Trypsin, a protease enzyme can hydrolyze the peptide bond only for which the surrounding groups are basic amino acids such as lysine and histidine etc.
4. Linkage specificity
The specificity of enzyme for the linkage or bond that they hydrolyze is termed as linkage specificity. Linkage specificity is sometime considered same to group specificity. However, in group specificity the enzyme considers both the type of bond/linkage and the surrounding groups of that linkage as well. For example, the protease enzyme for protein digestion hydrolyzes the
5. Optical specificity
Also termed as the stereo-specificity of enzymes. the enzyme show specificity not only for the substrate but also towards its optical configuration. This type of specificity is considered as the highest level of enzyme specificity.
The L-amino acid and the D-amino acids have different optical configuration so the enzyme such as oxidase are specific either for L-form or for D-form of amino acids.
Similarly the α-glycosidic bond of starch and glycogen can be hydrolyzed by the action of α-amylases and the β-glycosidic bond can be hydrolyzed by β-glycosidases.
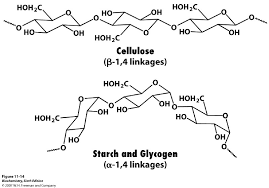
“Cellulose and starch has differ type of linkages. This is the reason that amylase enzyme of human body cannot digest cellulose due to its β-1,4 linkage.”
The amylase enzyme of human body cannot digest the cellulose because cellulose has β-1,4 glycosidic linkage and the enzyme can only break α-1,4 linkage of glycogen and starch.
6. Geometrical specificity
In this type of specificity, a single enzyme can perform catalytic activity on different types of substrates when they share same molecular geometry. This is low level of enzyme specificity.
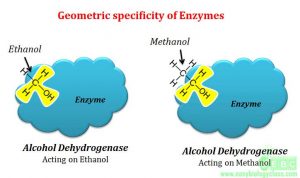
“Diagrammatical representation of geometrical specificity of enzymes”
The examples are alcohol dehydrogenases that have the ability to oxidize ethanol as well as methanol to yield aldehyde products. This is because both the alcohols share same molecular geometry.
7. Co-factor specificity
The non-protein components of enzyme that are essential for enzyme functions are co-factors. Some enzyme classes show specificity of the co-factor that is attached to them. The enzyme will work properly only in presence of specific co-factor and substrate molecules. Absence of specific co-factor can result in inactivation of enzymes even if the substrate is available.
For example, pyruvate carboxylase enzyme that converts pyruvate to oxaloacetate is biotin dependent enzyme.
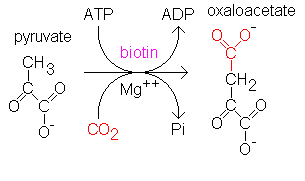
“Action of Biotin dependent pyruvate carboxylase enzyme”
On the basis of all these types of specificity patterns of enzyme, the functions of enzymes are also specific for a particular biochemical reaction.
Bottom line
Specificity of enzyme varies with particular enzymes and with reference to reaction and its substrate. With specific enzyme and substrate combination, the rate of reaction is ideal to proceed to maintain the cellular biochemistry.