National Institute of Health NIH has started the phase 1 trials for yet an investigational vaccine for treatment of coronavirus disease. This is in fact a significant breakthrough in research and development of vaccine against COVID-19. Researchers at National Institute of Allergy and Infectious Disease NIAID’s Vaccine Research Center, in collaboration with Biotechnological company Mederna, were able to quickly develop mRNA-1273 vaccine for COVID-19 treatment. Moreover, the prior studies and research about Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) are proved to be helpful for this vaccine development.
According to the latest (13 April, 2020) situation report by World Health Organization (WHO), there are 1 773 084 confirmed cases of COVID-19 (76, 498 new cases). And the death toll jumps to 111 652 deaths (5,702 new). Research and lab trails are being performed to develop a vaccine against SARS-COV-2 strain, or the coronavirus disease.
Investigational vaccine and clinical trials
Researchers at NIAID’s Vaccine Research Center in collaboration with biotechnological company Mederna have developed this vaccine named mRNA -1273 for COVID-19 treatment. The US National Institute of Health NIH has started the Phase 1 trials of this investigational vaccine against novel coronavirus.

The novel coronavirus is RNA virus, spherical in shape and it has protein spikes protruding from the surface. This crown-like virus attaches itself on the receptors surfaces of human cell. The virus particularly inhibits and attacks the respiratory tracts causing mild to severe pneumonia-like symptoms.
Phase 1 trials for mRNA-1273 vaccine for COVID-19
According to reports by NIH, the phase 1 clinical trials for mRNA-1273 have begun at Kaiser Permanente Washington Health Research Institute (KPWHRI) in Seattle. The participants of the study are young adults of age group 18-55 years.
The vaccine mRNA-1273 is intended to act against SARS-CoV-2 which encodes for a prefusion stabilized form of the S-protein (Spike protein of novel coronavirus). This vaccine is based on genetic sequence of mRNA (messenger RNA) molecules derived from the novel coronavirus strain. The vaccine is expected to direct the body’s cells to express a virus protein. Thus, it can elicit a robust immune response. The mRNA-1273 vaccine for COVID-19 has shown promising effects in animal’s models. Now, the trails have begun for human.
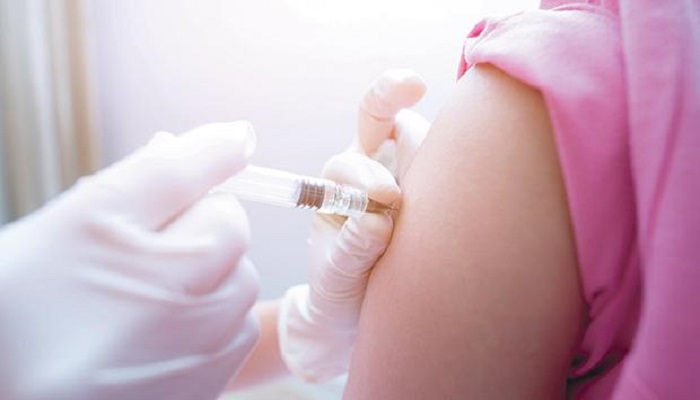
The trials are designed to investigate the effectiveness of mRNA-1273 vaccine for COVID-19. These trials will evaluate the vaccine safety as well as the immunogenicity of different doses. Initially, researchers have selected three dose levels of mRNA-1273 vaccine for COVID-19, 25 μg, 100 μg and 250 μg. The vaccine is scheduled in two doses with a gap of 28 days.
The project director Dr. Jackson at NIAID stated:
“This work is critical to national efforts to act in response to the threat of this emerging virus. “We are ready to conduct this important trial because of our experience as an NIH clinical trials center since 2007.”
Participant’s follow up
On the other hand, Mederna Company plans to have 45 healthy adults for the early-stage trial. Moreover, the participants of this study will be followed or monitored through 12 months after the date of the second vaccination. Participants will be asked to return to the clinic for follow-up visits between vaccinations. Furthermore, a protocol team will keep track of vaccination symptoms such as inflammation or soreness after injecting vaccine. Blood sample analysis and laboratory tests will be performed in order to detect and measure the immune response to the experimental vaccine.
So hopefully these trails will go successful so that the world can get rid of this pandemic. Until then, be very careful, take precautions and maintain a safe distance to be safe from this coronavirus disease.
Way cool! Some very valid points! I appreciate you
writing this article plus the rest of the site is also
really good.