COVID-19 latest (30-April-2020) situation report by World Health Organization (WHO);
- Confirmed cases: 3,181,642
- Confirmed deaths: 224,301
- Areas / country/territories affected: 215
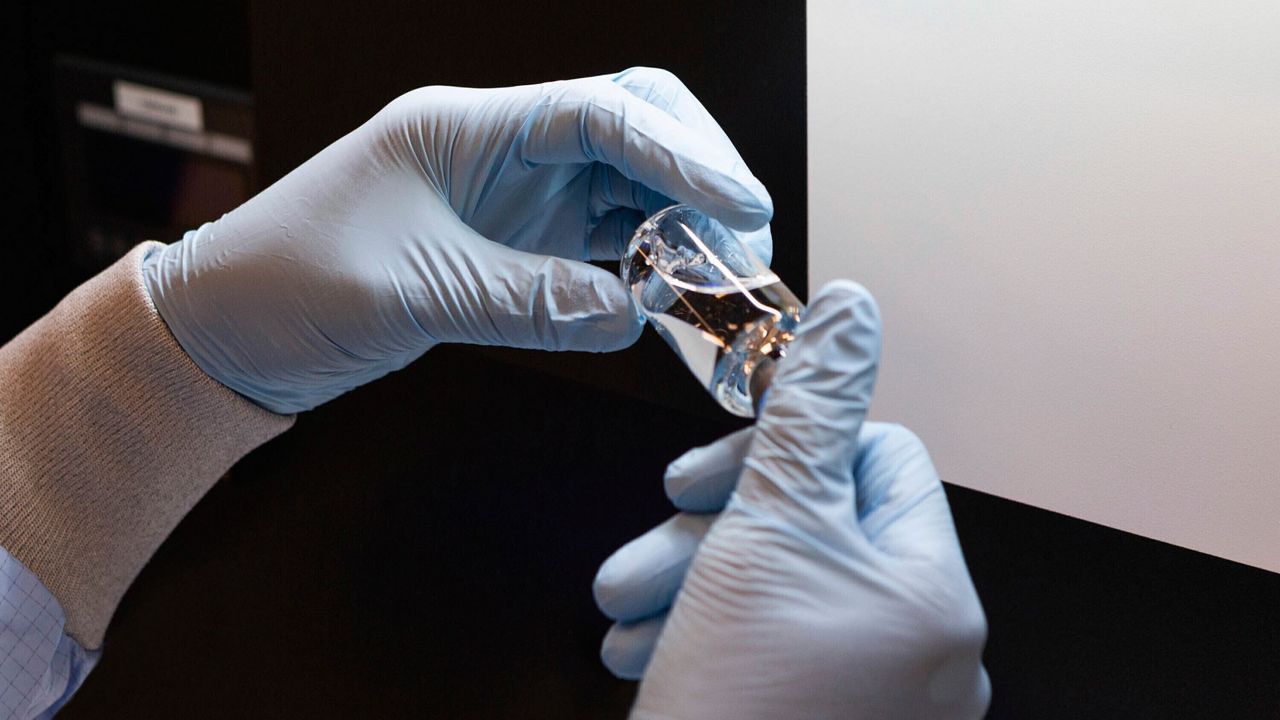
COVID-19 continues its killing spree across the globe. At the same time, the researchers and medicine companies are working tirelessly to develop a vaccine or some treatment against SARS-COV-2 virus. A pharmaceutical company, Gilead Sciences, Inc. has announced the results of phase-3 trial of an antiviral drug remdesivir against SARS-COV-2 or COVID-19. This antiviral drug might be effective against novel coronavirus or SARS-COV-2. On the other hand, the drug is still at investigational phase and not been approved yet by the US Food and Drug Administration (FDA) or any other authority for the treatment of COVID-19.
Phase-3 trials for remdesivir against SARS-COV-2
The phase-3 trials involved 5 days and 10 days dosing duration of remdesivir drug for COVID-19 patients. The results demonstrated that patients receiving a 10-day treatment course of remdesivir showed similar improvement in symptoms as those given a 5-day treatment course.
Chief medical officer Merdad Parsey of Gilead Sciences stated;
“Unlike conventional drug development, we are trying to evaluate an investigational agent alongside an evolving global pandemic. Multiple related studies are helping inform whether remdesivir is a safe and effective treatment for COVID-19. And how to best utilize the drug.’
The results of this study complement the data from a similar study on effectiveness of remdesivir performed by National Institute for Allergy and Infectious Diseases.

What is remdesivir?
Remdesivir is an investigational or experimental antiviral drug produced by the US pharmaceutical company Gilead sciences Inc. Originally, the drug was expected to be effective treatment for the Ebola virus and even showed promising results in lab trials.
In fact, remdesivir is a prodrug of a nucleotide analogue. The drug undergoes metabolic changes at intracellular level and develops an analogue of adenosine triphosphate which further inhibits viral RNA polymerases. This antiviral drugs has broad-spectrum activity against members of several virus families, including filoviruses (such as Ebola) and coronaviruses (including both SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV). In non-clinical lab trails, the drug has shown prophylactic and therapeutic efficacy against these corona viruses.
Study published in The New England Journal of Medicine for evaluating the effectiveness of remdesivir suggests promising effects of remdesivir against covid-19. Furthermore, the in vitro testing has also shown effectiveness and activity of this remdesivir against SARS-CoV-2. The drug appears to have favorable clinical safety profile.

The results of another study suggest otherwise!
The results of another study published in The Lancet Journal did not came out as expected. The study evaluated the effectiveness of remdesivir against COVID-19. The clinical trials suggest that intravenous remdesivir did not significantly reduce the time to clinical improvement, mortality, and viral load. Furthermore, the study reveals some of the expected adverse side effects. The participants showed side effects including hypoalbuminaemia, hypokalaemia, anaemia, and increased total bilirubin.
Although remdesivir showed strong antiviral effects in pre-clinical models of coronavirus infection, it did not provide expected results in clinical trials. The drug did not result in significant reductions in SARS-CoV-2 RNA loads or sputum specimens or in upper respiratory tract.
In addition to this, there is need to be early application of such investigational drugs before the symptoms get worse. Novel coronavirus replication is of short-duration and lung pathology appears rapidly unlike the other human infections.
Will it work?
Hopes are high for rapid development of some vaccine or drug against covid-19. Remdesivir is one of these investigational antiviral drug which is expected to be effective against SARS-COV-2. Till date, this drug is not yet licensed or approved anywhere globally. Furthermore, there is inconsistent data about the safety and effectiveness of remdesivir for COVID-19.
However, these studies are significant and helpful to evaluate the safety and effectiveness of any investigational drug or medicine. We really hope that these clinical trials and ongoing researches for developing a treatment for COVID-19, turn out as expected. Till then, we must take precautionary measures to keep ourselves safe and to contain the spread of COVID-19.